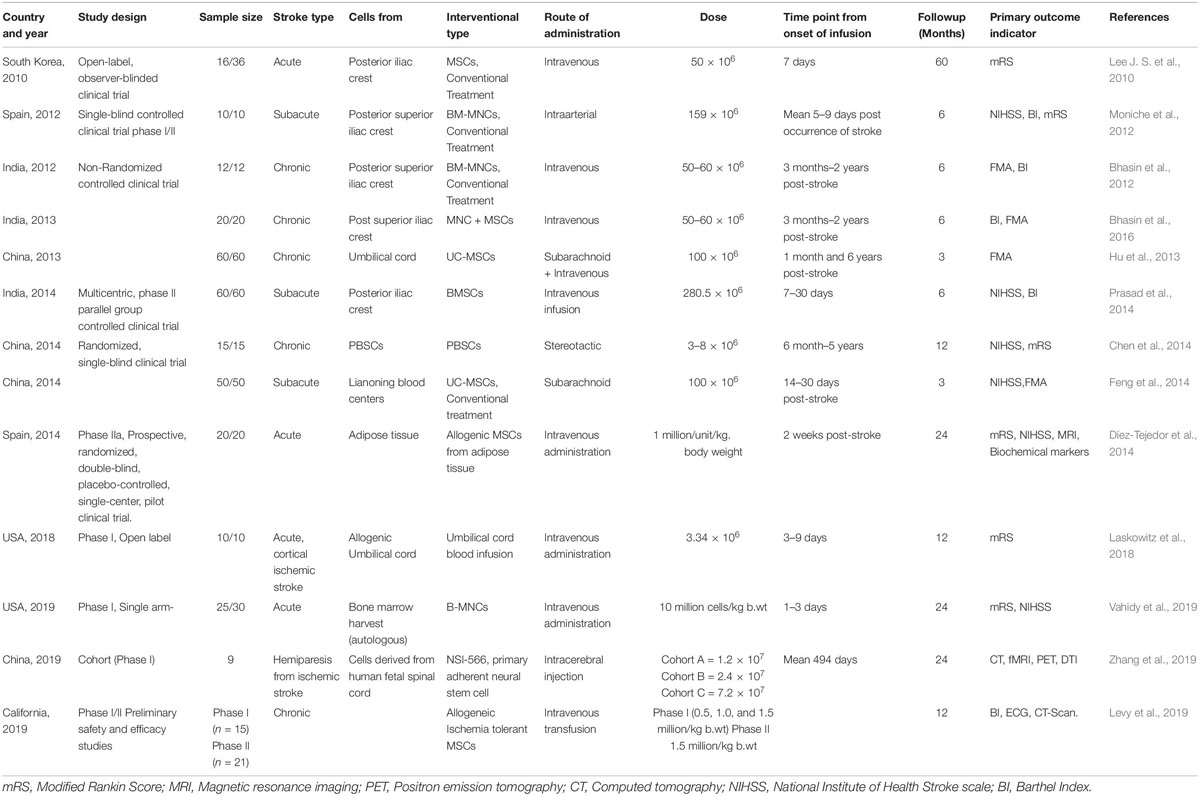
Adipose tissue-derived stem cells (ADSCs) are reported to have several advantages over bone marrow-derived stem cells. Both adipose tissue and umbilical cord are promising sources of MSCs. These other sources of MSCs outside bone marrow or allogeneic MSCs from young donors may address the apparent decrease in the number and function of bone marrow MSCs in aged persons. This has resulted from our current understanding of the characteristics of different sources of MSCs. These findings suggest the importance of the aging/rejuvenation of donor cells to the neurogenic potential of stem cell therapy.Īlthough bone marrow-derived MSCs are most commonly used, other sources of MSCs, such as adipose tissue or the umbilical cord, are increasingly utilized in clinical practice. On the contrary, age-related changes could affect certain biological features of bone marrow MSCs, resulting in decreased proliferation and paracrine functions as well as increased senescence and apoptosis, which may decrease the neurogenic potential of MSCs. 
One recent study showed that secreted factors from the young stem cell niche rescued the numbers of NSC colonies derived from old-age subependyma, and enhanced NSC proliferation in vivo in aged animals. In addition, NSCs in aged brains could be activated by application of “younger” stem cells. Although the number of NSCs decreased with age in the human brain and basal neurogenesis was impaired in the subgranular and subventricular zone of aged animals, the degree of neurogenesis after stroke was similar in young and old animals. However, stroke-induced neurogenesis has been observed in stroke patients in their 60s and 70s. An attenuation of the regenerative potential of stem cells in aged patients with stroke could result from aging in either the donor cells (e.g., bone marrow stem cells) or the recipient cells (e.g., NSCs in the innate neurogenesis system of the brain). The neurorestorative potential of MSCs may be limited in the elderly who have a limited number of neural stem/progenitor cells (NSCs) and bone marrow MSCs, who are unable to receive rehabilitation therapy, and those with extensive damage to the subventricular region. MSCs are heterogenous and contain many different types of progenitor or stem cells, in terms of growth, trophic support, and differentiation potentials. on behalf of AlphaMed Press.Third, adult stem cells may have inherited limitations. Stem Cells Translational Medicine published by Wiley Periodicals, Inc. Stem Cells Translational Medicine 2019 8:983&988.īasic science Cerebral ischemia Clinical Regenerative medicine Stem cell transplantation Translation. The laboratory and clinical experience with MSC therapy for stroke may guide the future translational research on stem cell-based regenerative medicine in neurological disorders. Echoing the Stem Cell Therapeutics as an Emerging Paradigm for Stroke and Stroke Treatment Academic Industry Roundtable call for an NIH-guided collaborative consortium of multiple laboratories in testing the safety and efficacy of stem cells and their derivatives, not just as stand-alone but preferably in combination with approved thrombolytic or thrombectomy, may further increase the likelihood of successful fruition of translating stem cell therapy for stroke clinical application. Two recently concluded stroke clinical trials on mesenchymal stem cells (MSCs) highlight the importance of strict adherence to the basic science findings of optimal transplant regimen of cell dose, timing, and route of delivery in enhancing the functional outcomes of cell therapy. Although safety of the grafted cells has been overwhelmingly documented, efficacy has not been forthcoming. Four decades of preclinical research demonstrating survival, functional integration, and behavioral effects of transplanted stem cells in experimental stroke models have provided ample scientific basis for initiating limited clinical trials of stem cell therapy in stroke patients.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
